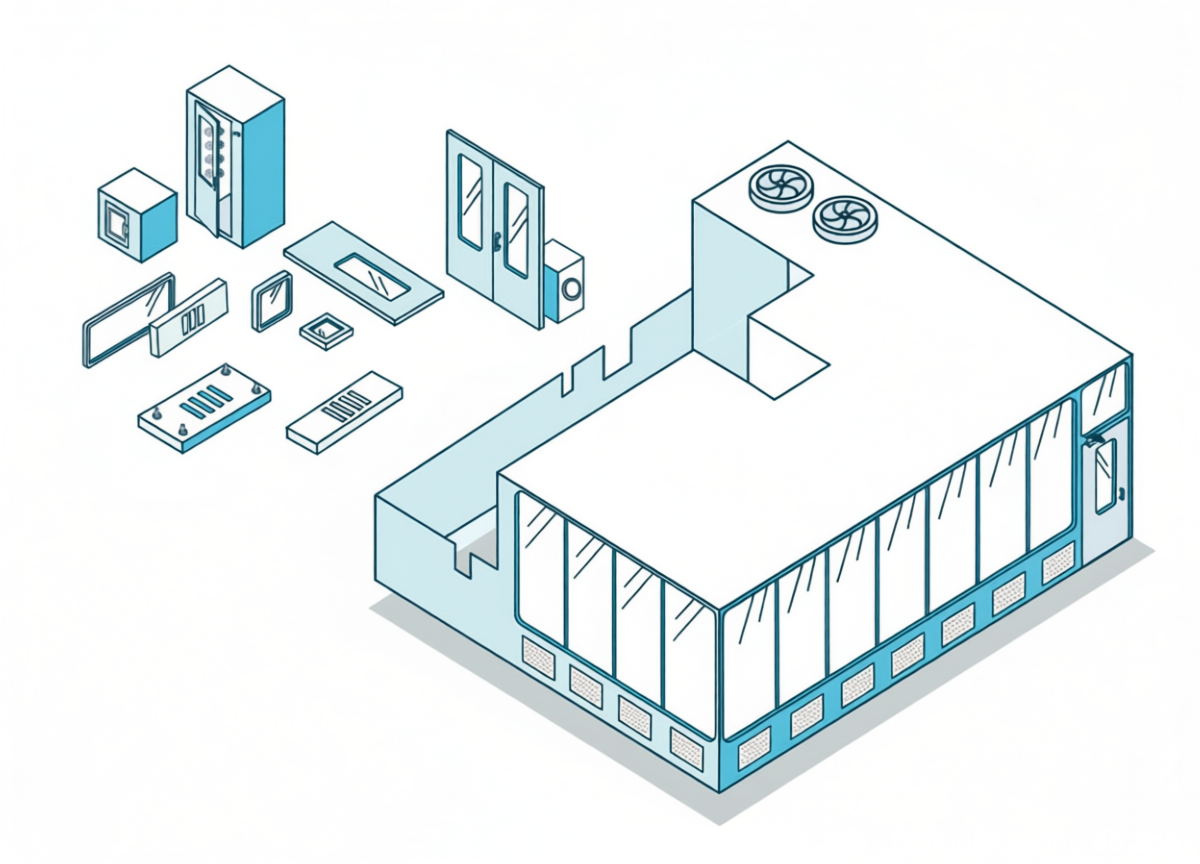
GMP-compliant cleanroom solutions for the pharmaceutical industry in Germany
ISO 7 / ISO 8 modular cleanroom systems | CE certified | EU GMP Annex 1
✔ Modular design
✔ Integrated HVAC
✔ Validation-ready
Are you facing these challenges in pharmaceutical production?

Strict EU GMP Annex 1 requirements
Precise pressure cascades & air exchange rates
Control of particles and germs
HVAC systems with validation requirements
Short project durations for new construction or expansion
“Technical expertise for the highest cleanroom standards”
― DERSION

-
 Layout optimization for production flow
Layout optimization for production flow Material and personnel separation
Material and personnel separation Documentation support for IQ/OQ
Documentation support for IQ/OQ Risk analysis according to GMP
Risk analysis according to GMP
“International Pharmaceutical Cleanroom Projects”
― DERSION







“Start your GMP cleanroom project today”
-
1. Free technical consultation / 2. Tailor-made offer / 3. Layout planning included