Industry Background & Pain Points
High sterility requirements:
Microbial and particle contamination must be avoided during drug preparation.
High compliance pressure:
Stringent standards such as FDA, EU GMP and USP <797> need to be met.
Difficult expansion:
Existing facilities are difficult to meet the needs of business growth.

Why Pharmacies Need USP-Compliant Cleanrooms?
Pharmacies in the United States are required to follow USP and USP standards for compounding sterile and hazardous drugs. These regulations aim to ensure the safety of patients and healthcare workers, minimize contamination risks, and control exposure to hazardous substances.
Common pharmacy challenges:
- Maintaining ISO-classified environments
- Preventing cross-contamination between hazardous and non-hazardous areas
- Managing air pressure differentials and airflow
- Providing proper gowning and material transfer procedures.
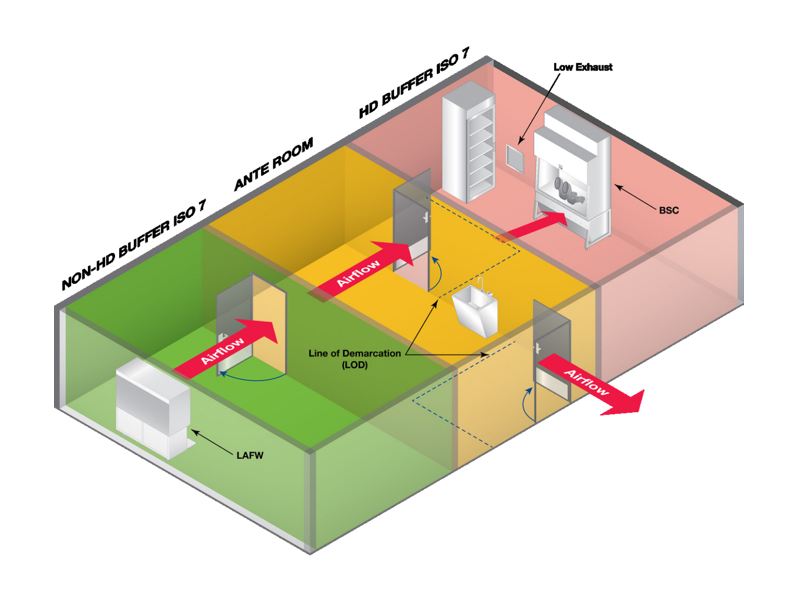
Cleanroom Zoning Overview
A USP-compliant cleanroom typically includes the following functional areas:
-
Gowning area for donning personal protective equipment (PPE)
-
Clearly marked dirty vs. clean zones on the floor
-
Hand washing sink located ≥1 meter from the entry door
-
Eye wash station near hazardous areas
-
Serves as a transition zone to both non-HD and HD rooms
-
Positive pressure relative to the Ante Room
-
Equipped with Laminar Airflow Workbenches (LAFWs)
-
HEPA-filtered air; supports sterile preparation of non-HD drugs
-
Negative pressure relative to adjacent rooms
-
Exhaust air is non-recirculated and vented outside
-
Contains Class II Type B2 Biological Safety Cabinets (BSCs)
-
Supports both HD and non-HD rooms
-
Stable temperature and humidity environment
-
Houses air handling units and support equipment
-
For segregated storage of hazardous drugs
-
Access from the ante room in the same airflow direction
Flow & Layout Principles
neutral/ante →Clean areas → Dressing Area → Buffer → Sterile preparation area
What is a GMP clean room?
In the pharmaceutical sense, a clean room refers to a room that meets the GMP specifications defined in the GMP sterility specifications (i.e., Annex 1 of the EU and PIC/S GMP Guidelines, as well as other standards and guidelines required by local health authorities). It is a combination of engineering, manufacturing, completion, and operational controls (control strategies) required to convert a normal room into a clean room.
According to the relevant standards of FDA agencies, they have established strict and precise regulations for pharmaceutical manufacturers in the pharmaceutical industry. Good Manufacturing Practices (GMP) for the manufacture of sterile pharmaceutical products are designed to ensure that drugs are safe and contain their claimed ingredients and quantities. These standards aim to reduce the risk of microbial, particulate, and pyrogen contamination. This regulation, also known as current good manufacturing practices (cGMP), covers production processes, quality control, packaging, personnel, and GMP facilities.

Matching GMP Grades to Pharmaceutical Operations

- Grade A: A localized zone for high-risk operations like aseptic filling and sterile connections. Typically achieved using a Laminar Air Flow (LAF) hood or isolator, situated within a Grade B background.
- Grade B: The background environment for a Grade A zone in aseptic preparation and filling.
- Grade C: Used for carrying out less critical stages in sterile product manufacturing or for terminal sterilization processes.
- Grade D: A preparatory area for less critical steps, such as washing equipment or handling components before sterilization.
Core Principles of Compliant Cleanroom Design
A compliant design integrates multiple elements to create a holistic contamination control strategy.
- Airflow and Pressure Cascades The single most important design element is control over air.
- Airflow: Unidirectional (laminar) flow is used in Grade A zones to sweep particles away from the critical process. Non-unidirectional (turbulent) flow is used in lower-grade rooms to dilute and remove contaminants.
- Pressure Differentials: A cascade of positive air pressure is maintained, with the highest pressure in the most critical area. This ensures air flows from cleaner to less clean areas, preventing the ingress of contaminants. A typical pressure differential between adjacent rooms is 10-15 Pascals.
Layout, Flow, and Finishes The physical layout must support logical process flows for personnel, materials, and waste to prevent mix-ups and cross-contamination.
- Materials: All surfaces must be smooth, non-shedding, non-porous, and resistant to repeated cleaning and sanitization agents. Common materials include epoxy-coated floors, uPVC or HPL wall panels, and integrated coving between walls and floors.
- Layout: The design must include dedicated airlocks for personnel (gowning rooms) and materials (Material Airlocks - MALs) to create buffers between different classification zones.

Critical Technical Systems: HVAC and Monitoring

- Pharmaceutical HVAC Systems The Heating, Ventilation, and Air Conditioning (HVAC) system is the "lungs" of the cleanroom. It is responsible for delivering the correct quantity and quality of air, controlling temperature and humidity, and maintaining pressure cascades. Key components include dedicated Air Handling Units (AHUs), multi-stage filtration (including terminal HEPA/ULPA filters), and sophisticated control systems.
Environmental Monitoring Systems (EMS) A modern cleanroom requires a continuous or frequent monitoring system to provide real-time data on its state of control. This includes:
- Non-viable particle counters
- Viable (microbial) air samplers
- Pressure differential gauges
- Temperature and humidity sensors
Our GMP Pharmacy Cleanroom Solutions

Customized design:
Customize cleanroom layout and functions according to pharmacy needs.
Compliance guarantee:
Cleanrooms comply with FDA, EU GMP and USP <797> standards.
Flexible expansion:
Modular design supports later expansion and upgrade.
